Solution
UDI Identification Comprehensive Solution – One-Stop Service
April 12, 2022
With the introduction of the Regulations on the Unique Device Identification (UDI) System by the National Medical Products Administration (NMPA), the medical device industry has officially entered an era of traceability management with unique identification for each product.
The UDI (Unique Device Identification) system enhances the ability of manufacturers and regulatory authorities to trace and manage medical devices throughout the supply chain. It enables the rapid and efficient recall of products with potential safety risks. For domestic medical device manufacturers, understanding UDI implementation and mastering UDI coding technology at an early stage is essential for aligning with international standards and integrating into the global UDI system.
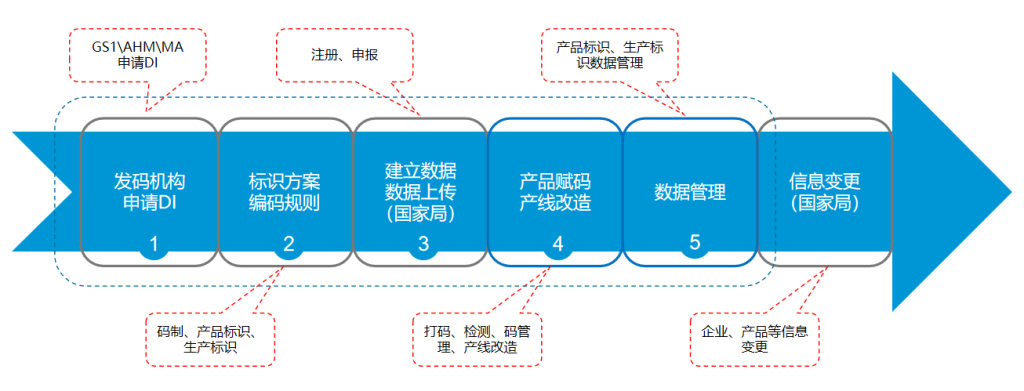
I. Overall UDI Implementation Process
The UDI system follows a unique coding structure that assigns each product a distinct identity, ensuring traceability of its origin and movement. This system also standardizes and regulates all stages of the production process, providing a comprehensive approach to medical device management.
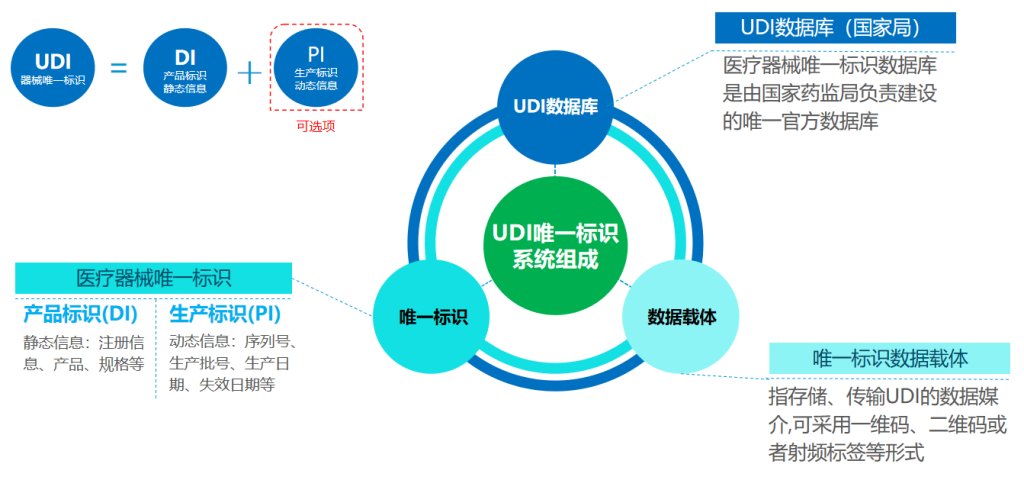
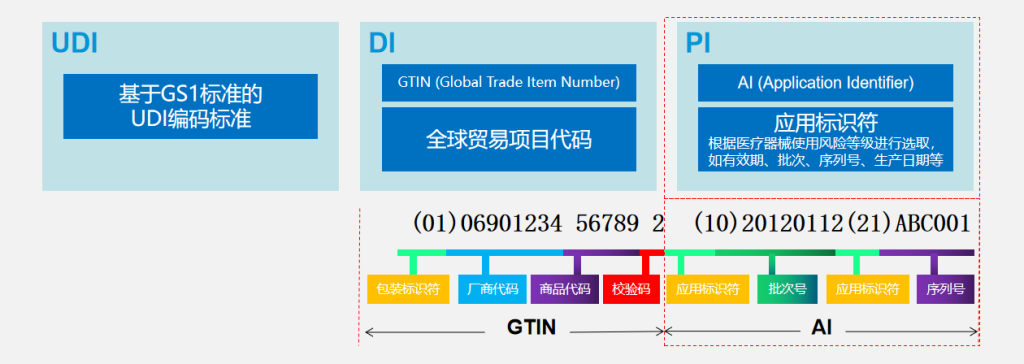
II. UDI Code Structure
III. UDI Coding Hardware
GaoYin Industrial provides a comprehensive range of UDI coding hardware solutions, ensuring compliance with various packaging types and production lines. Our solutions support coding applications for cartons, paper boxes, metal, plastic, and film packaging materials, helping enterprises implement customized and regulatory-compliant UDI marking systems.
Our UDI coding hardware ensures:
- High-precision marking for different surfaces and materials.
- Seamless integration with existing production lines.
- Reliable traceability for regulatory compliance.
With our complete UDI hardware solutions, enterprises can efficiently assign, manage, and track UDI codes while ensuring smooth implementation across all packaging and production environments.

IV. UDI Compliance One-Stop Solution
1. UDI Code Editing and Printing
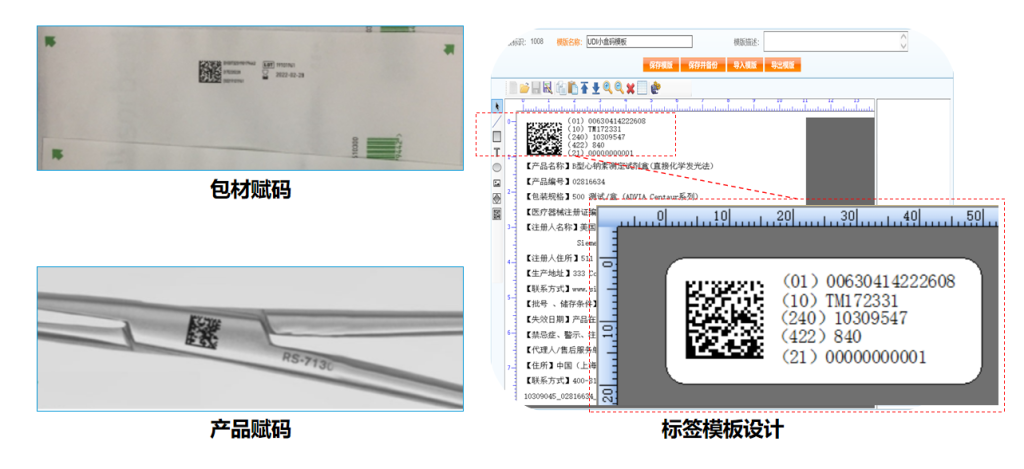
2. Providing Two Solutions Based on Different Enterprise Needs

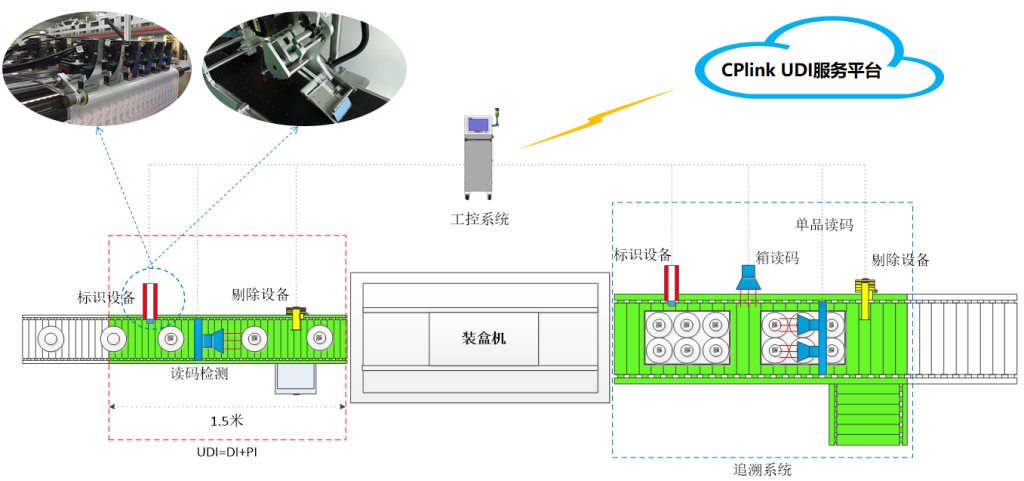
V. UDI System Integration Software Architecture
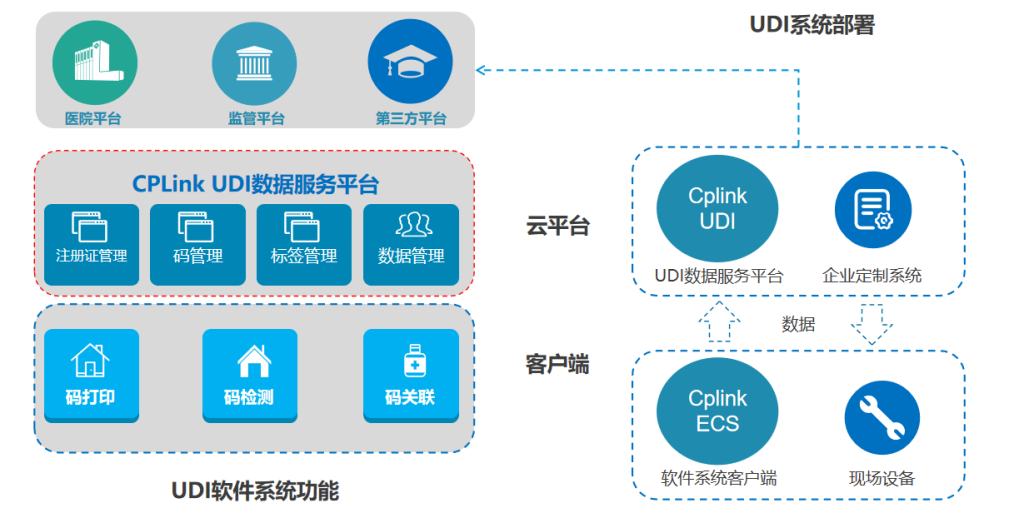
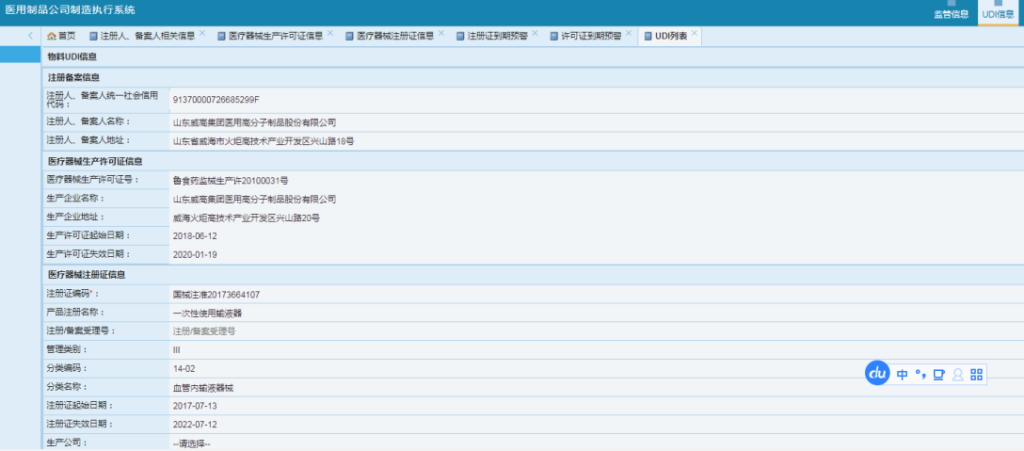
VI. Showcase of Some Service Clients
